Cardizyme
Catabody Cardizyme For Aging
Investment Rationale
-
US Market: Systemic TTR amyloid deposits in the heart, joints nerves, joints and other locations are is a major cause of aging. Cardizyme may garner a significant share of the age-associated transthyretin (TTR) amyloid disease market (>4.5 Mil patients).
-
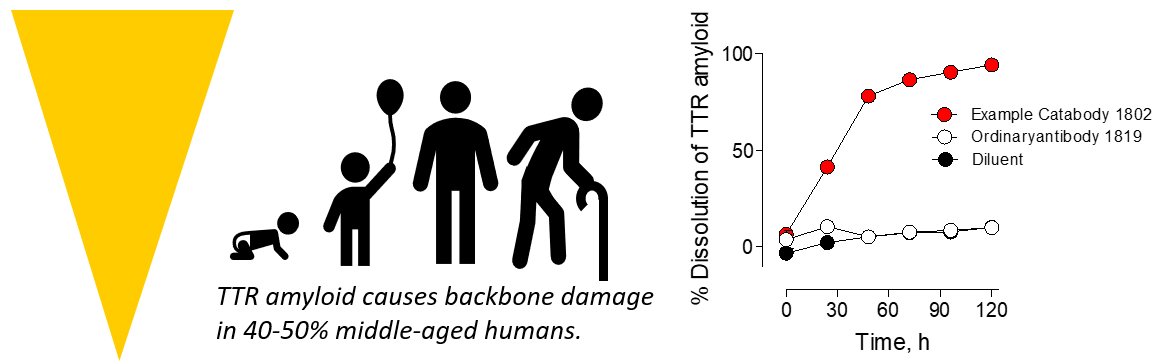
Medical Advantages: Only high efficacy drug candidate that targets, destroys and dissolves the cause of disease, misfolded TTR without interfering functions of properly folded TTR. Unique mechanism. Destroys particulate and soluble misfolded TTR completely at nanomolar concentrations. Works ex vivo for dissolution of misfolded TTR amyloid in human tissues. Unlike competitor drugs, has little or no prospect for inducing inflammation or interfering with vision and sugar/lipid metabolism.
-
Cardizyme Prioritization: Cardizyme targets the singular disease cause, misfolded TTR, and is our priority product with shortest route to market. Milestone-driven Cardizyme development plan will offer exit opportunities at increased valuation for both long-term and relatively short-term investors.
-
Cardizyme Development: Preclinical milestones are: Define FDA-approvable efficacy and safety criteria at a pre-IND meeting; optimize Cardizyme expression by a renewable cell line; conduct chemistry and manufacturing control tests; show ex vivo TTR amyloid removal from various human tissues; and show consistent Cardizyme tissue delivery and safety in animals (8 months). We anticipate completing Cardizyme Phase 1 trials in patients with age-associated and familial TTR amyloid disease 30 months after capitalization. In addition to hitting the safety milestone, we want to see at least an initial signal for drug efficacy.
